By Charlie Powell, College of Veterinary Medicine
PULLMAN, Wash.—Most, if not all, infections and diseases in animals and people are met with some level of the body’s own inflammatory response. Sometimes this inflammatory response crosses a line from being protective and useful to becoming debilitative or even deadly.
Scientists in Washington State University’s College of Veterinary Medicine and College of Pharmacy and Pharmaceuticals Sciences (CPPS) have discovered an important step in the progression of the body’s inflammatory response that may soon lead to effective management and treatments. The team’s paper was published Monday in the prestigious journal, Nature Communications.
“Inflammation is vital for life, without it you will die,” said Professor Santanu Bose with WSU’s Department of Veterinary Microbiology and Pathology. “But inflammation can also debilitate or kill the very organism it is trying to save. Diabetes, Alzheimer’s disease, atherosclerosis, arthritis, cancer, pneumonia, and other infections are all examples where even after the benefits of inflammation have been attained, it is the continued, seemingly uncontrolled inflammation that makes things much worse, even deadly.”
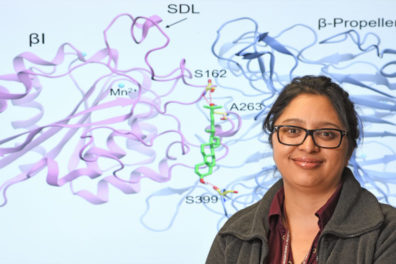
The collaborative team effort was led by Bose along with pharmaceutical sciences Assistant Professor Senthil Natesan in CPPS, and the former director of WSU’s School of Molecular Biosciences, Professor Jonathan Jones. [Of the 11 scientists contributing to the work, two were members of the CPPS faculty, Senthil Natesan postdoctoral research associate Jeevan GC (pharmaceutical sciences, Natesan lab).] The collaborative team’s work led to the discovery of a naturally-occurring modified form of cholesterol that binds to key cell surface proteins called integrins. Once bound, the complex acts to signal surrounding cells at the site of an infection or during metabolic insult to induce inflammation.

“What we found was that when a white blood cell called a macrophage encounters a foreign organism, say a virus, a gene in the cell turns on and produces a type of cholesterol,” Bose explained. “In our case we used an influenza A virus and respiratory syncytial virus as our models. This cholesterol then converts into another form of the lipid named 25-hydroxycholesterol or 25HC for short.
“Once produced, 25HC then leaves the cell and encounters other nearby macrophages where a receptor protein on the surface called an integrin is waiting, sort of like an antenna that waits to receive a radio signal. Once 25HC binds to the integrin it amplifies the inflammatory response.

“This is the first time scientists have found a lipid acting in this manner to amplify inflammation.”
The research group is also excited about the potential of this discovery in terms of developing anti-inflammatory therapeutics.
“This collaborative study shows the increasingly essential and complementary role of computer simulations in understanding complex, molecular level, biological processes,” explained Natesan. “One of the most novel and exciting findings of this study is the binding site through which 25HC interacts with the integrins. This seemingly druggable binding site opens up new opportunities to discover small molecule therapeutics to treat inflammation. Such an effort is currently underway.”

The study has also paved the way for future research directions in understanding immune cell biology.
“Integrin receptors are key regulators ensuring cells and tissues function normally,” said Jones. “Identification of this pathway provides a better understanding of how lipids affect integrin functions in both normal and diseased tissues. Our data might also explain why taking anti-cholesterol medications like statins actually decrease certain cancer risks since they inhibit integrins.”
Currently, the team is working closely with WSU’s Office of Commercialization to develop anti-25HC based anti-inflammatory therapeutic agents to combat various inflammatory diseases.
Bose gratefully says that the discovery would not have been possible without the National Institutes of Health funding support from his and Jones laboratories.
“Credit for this discovery, goes to my current and former graduate students and post-doctoral fellows from WSU and at the University of Texas,” said Bose.
Bose’s current graduate student Swechha Pokharel, is first author on the paper, and she worked equally closely on the project with Bose’s former graduate student Niraj Shil. Pokharel will earn her Ph.D. degree this coming fall.
“Swechha is to be commended for two remarkable things as a Ph.D. graduate student relative to this research,” said Bryan Slinker, dean of WSU’s College of Veterinary Medicine. “First, it is uncommon for a person who has not yet earned their doctorate to be first author on a paper in a journal as prestigious as Nature Communications. Second, her intelligence and warm, professional personality made working with a federally-funded, all WSU effort, that much more effective.”
“I am extremely grateful to Professor Bose,” said Pokharel of her mentor. “He recognized my willingness to learn and as a mentor he taught me how one brings together a large group of like-minded scientists readily willing to offer their expertise and direct their efforts for a shared discovery. I am blessed by this experience.”
A ghost from our past?
How did humans evolve to have an immune system that produces an inflammatory response that may kill them?
Professor Santanu Bose says, that’s an interesting question. He leans toward the questioner and asks with a smile, “Do you believe in residual spirits?”
The question is not one about the supernatural but more one referring to the evolutionary past of humans.
“This is pure speculation but, long before humans were civilized and we were wandering the plains or seashore in search of food, we may have eaten anything we came upon,” said Bose. “Imagine a dead animal. We ate the carrion and it was loaded with things that could make us sick or even kill us. Sometimes it probably did. But many times maybe, our immune system had this massive inflammatory response and sent all resources forth to kill off those pathogens and those humans survived to pass on their genes.
“No one knows for sure but maybe, our ability to do that has not been lost and now that we are not dependent on exposure to such things, our inflammatory process still stands ready? Perhaps what we call an overactive inflammatory response is coded in our genes and is one of the things that helped us survive as a species until now. For fun, one can think of it sort of like the residual spirits or ghosts in literature that inhabit a vessel and we are the vessel that carries them forward through time until we evolve away from it.”
Contact:
Charlie Powell, College of Veterinary Medicine, charlie_powell@wsu.edu, 509-335-7073